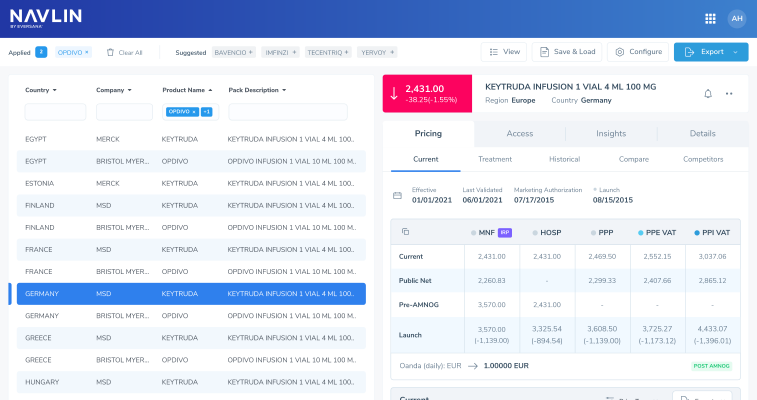
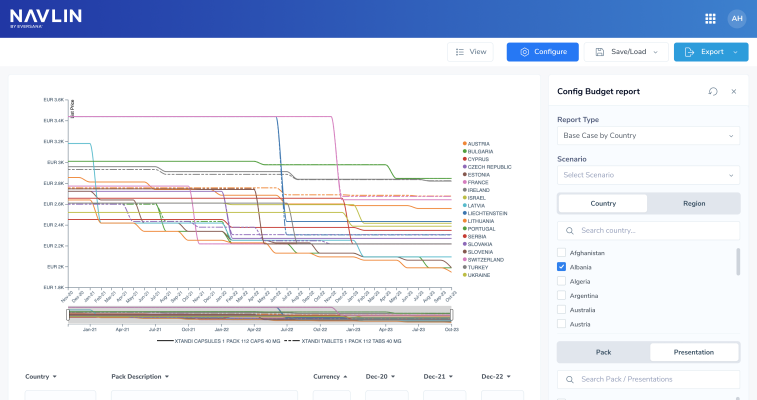
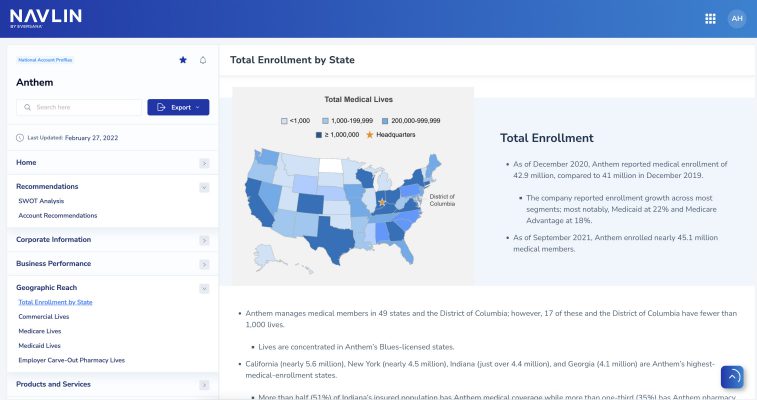
NAVLIN Daily NewsCheckout Today’s News
The National Healthcare Security Administration (NHSA) recently published its proposed work plan for this year’s adjustments to the National Reimbursement Drug List (NRDL) and the Commercial Insurance Innovative Drug List. The plan expands eligibility criteria for both lists and introduces flexibility for innovative drugsPrice control rules were also updated to link insurance payment standards with the list prices of competitively bid products, thus addressing notable price differences observed between list prices and reimbursement standards for some competitively priced products on the NRDLFurthermore, renewal rules were clarified, with a two-prong payment standard adjustment mechanism proposed for exclusive products for which agreements have expired after eight years, thereby alleviating past uncertainty about how contracts can be renewed after eight years and how prices will change after shifting to the standard list
The National Healthcare Security Administration (NHSA) published a tentative work plan for updating the National Reimbursement Drug List (NRDL) and the Commercial Insurance Innovative Drug List this yearThe plan proposes expanding eligibility to include drugs that have completed their technical review by the time the finalized work plan is issued. It also introduces NRDL eligibility for drugs that were on last year's commercial list, new generic products, and previously negotiated drugs that later failed the renewal processThe work plan opens the doors to the commercial list for new generics and exclusive products for rare diseases approved in the last five years or that have completed technical review

The European Parliament and Council negotiators have reached a provisional agreement on the Critical Medicines Act (CMA) aimed at reducing shortages of essential medicines, cutting the EU’s dependence on non-EU suppliers and strengthening pharmaceutical manufacturing inside the blocPublic Health Committee Chair Adam Jarubas (EPP, PL) said: "The Critical Medicines Act is closely linked to the pharmaceutical package, the missing piece of the EU's response to medicines shortages following the creation of the critical medicines list and the Critical Medicines Alliance. It implements the Pharmaceutical Strategy for Europe and marks a milestone for the European Health Union. It means greater medicines security for the Europeans, but it will also be an essential support for EU's reindustrialization and competitiveness"A Presidency debriefing on the outcome of the trilogue will take place on May 13, 2026. However, the provisional agreement still needs to be approved by both Parliament and Council before the new rules can enter into force
Last week, NAVLIN Daily brought you key market access developments from across the globe. Manufacturers discussing MFN/tariff agreements during Q1 2026 earnings calls shifted focus toward ex-U.S. reimbursement increases rather than U.S. policy changes, while the White House announced that it predicts $64.3B in federal and state savings from MFN In Europe, the HTACG granted Ipsen’s Ojemda (tovorafenib) the first-ever JCA approval under the joint HTA Regulation CMS extended the GENEROUS Model application deadlines for manufacturers and states, while Brazilian industry representatives raised concerns over pricing the newly implemented drug pricing regulation
China rolled out a sweeping plan to improve children’s healthcare through drug development incentives, faster marketing approvals, and streamlined reimbursement decisions. Local and foreign drugmakers benefit from the measures, with added perks such as expanded patent protection and market exclusivity periodsHowever, some areas of the policy aim to benefit the domestic industry, including measures to reduce reliance on foreign combination vaccines and relaxed rules for Chinese clinical dataOther specific aspects of the policy include expanded use of real-world evidence (RWE) and prioritized centralized procurement and entry onto the National Reimbursement Drug List (NRDL)
Torrent Pharmaceuticals has emerged as an early leader in India’s fast-growing generic semaglutide market, capturing nearly 38% market share within a month of launch“Torrent’s early lead appears to be driven by oral semaglutide,” Aasim Maldar, consultant endocrinologist and diabetologist at PD Hinduja Hospital, told The Economic Times“Many patients would prefer a tablet over an injection, given the choice,” he said
A new analysis published by White House economists predicts that the U.S. administration's voluntary deals with drugmakers will save the U.S. economy billions across all payer channels over the next 10 yearsOverall, the report predicts $64.3B in federal and state savings over the next 10 years stemming from applying MFN prices to state Medicaid programs and an estimated $529B in domestic savings across all markets (inclusive of commercial insurance) stemming from manufacturers offering all new drugs launched in the U.S. at prices comparable to those in other high-income countriesThe analysis also reiterates the administration's commitment to getting other countries to pay more for medicines, and highlights the recent U.S./UK deal as an example of what the U.S. is looking to "replicate" with other nations
Throughout recent Q1 2026 earnings calls, executives of companies that have struck Most Favored Nation (MFN)/tariff deals with the U.S. administration have demonstrated a shift in focus when it comes to MFN policy Instead of discussing U.S. policy developments and the potential implications of the U.S. administration’s proposed MFN pricing models (like GLOBE and GUARD), pharma leaders are homing in on efforts to increase reimbursement rates in ex-U.S. nations Keep reading for NAVLIN Daily’s round-up of the key policy and pricing-related conversations from this quarter’s earnings calls
The Danish Medicines Council has recommended four new drugs as standard treatment across oncology and rare diseases in April: Perjeta (pertuzumab), Rubraca (rucaparib), Loqtorzi (toripalimab) and polihexanide Two drugs were not recommended due to pricing and evidence concerns, while one oncology drug was placed on hold for further price negotiationsIn the same meeting, the Council adopted a new model that will enable access to PD-(L)1 inhibitors within 10 days of marketing authorization, replacing the previous 14-week individual assessment process
Last week, Germany made progress on the GKV reform bill, which will now move to the Bundestag for debate before final approval, and Japan decided to push ahead with FY2027 drug price cuts, despite MFN threats, while Brazil implemented the first overhaul of its drug pricing system in 20 yearsRegeneron signed 17th voluntary MFN deal with the White House, gaining relief from future pricing mandates and tariffs for three yearsIn Europe, Novartis’s Itvisma became the first ATMP undergoing the JCA process to receive a positive CHMP opinion, while speakers at the ABPI conference in London revealed that there are currently approximately 54 topics scheduled to enter the new MHRA-NICE aligned pathway