NAVLIN Daily NewsCheckout Today’s News
Last week, NAVLIN Daily brought you key market access developments from across the globe. The European Commission opened the third 2026 submission window for joint scientific consultations (JSC) under the EU HTA Regulation U.S. lawmakers introduced the Biotech Investment National Security Act of 2026, a proposal that would tighten oversight of U.S.-China biotech collaborations and investments on national security grounds Brazil approved new regulations that will allow CMED to impose stricter price controls on newly launched drugs and expand mandatory discounts in public procurement
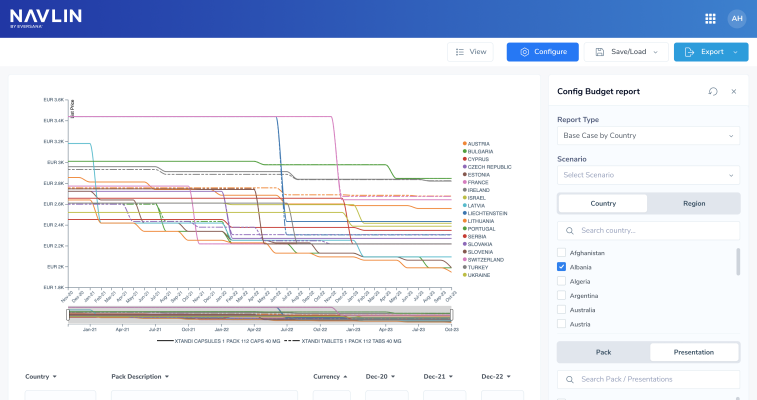
Spain’s proposed Medicines and Health Products Law has entered a crucial stage of review, introducing reforms that could significantly reshape pharmaceutical pricing and National Health System (SNS) financingThe draft legislation, currently under examination by the Council of State before its expected submission to Parliament, contains substantial revisions compared with the version previously released for public consultationAccording to Diario Farma, which has seen the new draft, the most significant change is the abandonment of the previously proposed “selected prices” model. In its place, the Ministry has developed a new pricing architecture that combines public financing mechanisms, reference pricing and a novel dynamic pricing system
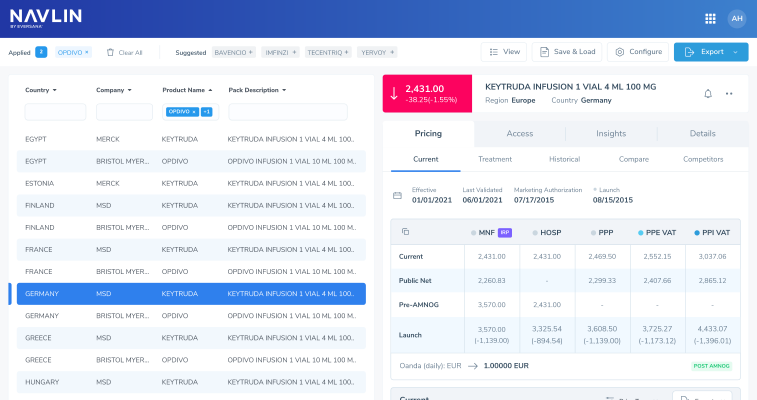
The Netherlands National Health Care Institute (ZIN) has advised the Minister of Health, Welfare and Sport (VWS) to place Novartis’s Itvisma (intrathecal onasemnogene abeparvovec) in the lock for expensive medicinesItvisma, indicated for patients aged two years and older with 5q spinal muscular atrophy, is expected to be priced at EUR 1.945 million per treatment, similar to ZolgensmaMedicines are placed in the lock if they meet either of two thresholds: an expected annual national cost of EUR 20 million or more for one or more new indications, or an annual per-patient cost of at least EUR 50,000 combined with a total annual cost of EUR 10 million or more
Michigan Reps. Debbie Dingell (D) and John Moolenaar (R) introduced a bill to tighten U.S.-China biotech investment regulations, citing national security concerns. The Biotech Investment National Security Act of 2026 would require the Treasury to scrutinize cross-border collaborations, targeting licensing deals between Chinese and U.S. companiesThe bill urges strict oversight of U.S. investments in China’s biotech sector, requiring defense and intelligence coordination to assess risks and a report on national security impacts. Critics argued that the bill is unnecessary and suggested streamlining U.S. clinical trials to compete globallyLawmakers are split, with Moolenaar warning of risky collaborations, while others highlight benefits like cheaper Chinese assets that can be developed to meet FDA standards
Details of Spain’s newly approved Royal Decree regulating the evaluation of health technologies (ETS) have been published in the Official State GazetteAs reported earlier this week, the Council of Ministers has approved the framework, in a move the government says will modernize healthcare decision-making and align the country with new European Union rulesA subtle modification appears in Article 15.1. As reported earlier this week, in the Ministry of Health’s own press release, it stated: “The purpose of the regulation is to establish guidelines for evaluating health technologies when these analyses—which will be mandatory but not binding.” However, the final BOE text removes the term “mandatory” entirely and states only that evaluations are “non-binding” and not compulsory
In an interview with NAVLIN Daily, Shalabh Gupta, Founder and CEO of Unicycive Therapeutics, explained the most pressing considerations in the current U.S. biotech landscapeAccording to Dr. Gupta, companies should consider payer evidence generation in the earliest stages of drug development: “You can never be too early, but especially by the time you get into the clinic or start the clinical trials”Dr. Gupta also reminded developers to understand their products’ “overall commercial viability" because a best-in-class profile isn't always enough to succeed. Sometimes, he said, "it comes down to the payer mix"
New Zealand’s Medsafe is officially adopting a new verification pathway to fast-track approvals of drugs already authorized by at least two recognized foreign regulatory agenciesThe pathway has been finalized after the agency incorporated feedback from an industry consultation period earlier this year. It will take effect on July 3Medsafe removed requirements regarding evidence of approved variations of the initial marketing authorization and redactions in assessment reports. It also extended the time window for companies to respond to information requests

Spain's Council of Ministers has approved the Royal Decree regulating the evaluation of health technologies (ETS), a regulation which aims to establish a comprehensive, transparent, and coordinated framework for analyzing medicines, medical devices, and other tools used in the National Health System (SNS)The Ministry of Health explains “the purpose of the regulation is to establish guidelines for evaluating health technologies when these analyses—which will be mandatory but not binding—are used to inform public decisions regarding funding, pricing, inclusion in the portfolio of services, changes to conditions of use, or the withdrawal of health technologies” The government has also introduced strict timelines for assessments. Clinical and non-clinical evaluations for medicines will each have a maximum deadline of 90 days, while non-pharmaceutical technologies must be assessed within 180 days. Where a joint clinical assessment has already been carried out by the EU, Spain must issue its national clinical report within 15 days
The European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) has recommended eight new medicines for approval, including 13 indication extensions in its May meetingAmong the recommendations are Boehringer Ingelheim’s Jascayd (nerandomilast) for the treatment of idiopathic pulmonary fibrosis (IPF) or progressive pulmonary fibrosis (PPF), and AstraZeneca’s Etcamah (camizestrant) for the treatment of adults with locally advanced or metastatic breast cancer with a specific mutation in the ESR1 geneMeanwhile, CHMP recommended an extension to the marketing authorization for Novo Nordisk’s Wegovy (semaglutide) for weight management to add a daily oral tablet as alternative formulation to weekly subcutaneous injections

Last week, NAVLIN Daily brought you key market access developments from across the globe. In Europe, the EFPIA released its annual W.A.I.T. Indicator report, showing that average patient access timelines for approved medicines across the EU increased to 597 days, compared with 578 days in last year’s report. Meanwhile, in the U.S., the White House expanded its DTP platform, TrumpRx, to include access to more than 600 generic medicines Greece approved legislation extending the “5/11” reimbursement criterion to medicines entering through the IFET early-access pathway while removing Germany and Italy from its reference country list, as Japan’s MHLW considered bringing the implications of the U.S. MFN policy to the Chuikyo for formal discussion NAVLIN Daily attended a slew of conferences last week, including ISPOR U.S., FT U.S. Pharma and Biotech Summit, and Pricing & Contracting USA 2026. At ISPOR 2026, Diane Munch highlighted that while the U.S. MFN policy remains a key source of uncertainty impacting global launch sequencing, China’s industrial development is becoming a more significant focus for investors